Founded in 1986 by Bob Urie, Mediplus began with a clear mission: to develop high‑quality urodynamic disposables through direct collaboration with UK clinicians, starting with Southmead Hospital in Bristol. This hands‑on approach established a culture of clinician‑led innovation that still defines the company today.
Over nearly four decades, Mediplus has grown from a small home‑based operation into a global leader in single‑use medical devices, with expertise spanning Anaesthesia, Urology, Gynaecology, Urodynamics, and Operating Room care. Today, the company exports to 40+ countries while remaining headquartered in High Wycombe, UK.
Our Mission: Advancing Patient‑Centric Medical Technology
Mediplus is committed to improving patient outcomes through innovative, minimally invasive medical devices developed in partnership with healthcare professionals worldwide.
Managing Director Emma Gray emphasises the company’s purpose:
“We develop products that reduce trauma for patients while improving ease of use for clinicians.”
Cutting‑Edge Medical Device Innovation
Key Historical Innovations
Mediplus has successfully brought more than 15 major innovations to market by working closely with urologists, anaesthetists, gynaecologists and surgical teams.
These include:
• CT3000 – the world’s first non‑invasive pressure‑flow study system for predicting bladder outlet obstruction.
• Mediplus IV administration sets – giving clinicians safe and flexible access to a range of infusion pump systems.
• S‑Cath™ System – a breakthrough suprapubic catheter insertion kit, awarded the Queen’s Award for Enterprise (Innovation) in 2017.
Latest Mediplus Innovations (2024–2025)
S‑Cath™ System Long (2025)
To meet increasing global demand for advanced suprapubic catheterisation tools, Mediplus launched the S‑Cath™ System Long, an extended‑length version of its award‑winning device. The system enhances clinician choice and supports complex procedures requiring additional length.
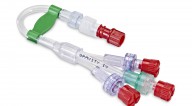
Revolutionary 5‑Way TIVA Set (SIVA 2024 Launch)
Mediplus unveiled the first‑of‑its‑kind 5‑Way Total Intravenous Anaesthesia (TIVA) Set, enabling clinicians to administer four drugs plus an infusion via a single integrated hub.
Benefits include:
• Streamlined anaesthesia workflows
• Fewer line connections
• Lower risk of leaks & blockages
• Anti‑siphon and anti‑reflux safety valves
• Colour‑coded Siamese tubing
• Reduced plastic usage for improved sustainability
This device represents a major advancement in anaesthesia safety and efficiency.
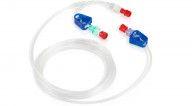
Peripheral IV Connectors
Peripheral IV connectors are designed to ensure optimal drug delivery for enhanced safety in ICU, HDU, orthopaedic and obstetric departments. These high-performance connectors (Chalfont, Coventry and High Flow) offer a wide range of benefits:
• Enables rapid emergency response through fast flow rate for rapid IV fluid delivery.
• Greater control through the integrated anti-siphon valve prevents free flow and back flow.
• Comfort and convenience with the removable bridge prevents the device becoming dislodged.
• J-loop design (Coventry and Chalfont Connectors) prevents kinking and occlusion of tubing for unimpeded flow.
• Single, double or triple infusion options available.
POPY™ Pessary Innovations
POPY™ is recognised as the world’s first silicone shelf pessary, marking a major innovation in the management of pelvic organ prolapse (POP).
• The flexible silicone design decreases the likelihood of vaginal erosion, a common issue with harder pessaries.
• Added five new sizes, expanding the range to 11 sizes, available in short and long stem configurations.
• This supports a more precise, personalised fit, enhancing effectiveness across a wider patient population.
Capnomask™ EtCO2 Monitoring
Capnomask is fast and reliable, enabling continuous respiratory monitoring in spontaneously breathing patients - particularly useful during:
• Monitored anaesthesia care (MAC)
• Procedural sedation
• Post‑anaesthesia recovery
Capnomask is a specialised oxygen face mask that includes a built‑in sampling port for capnography, allowing clinicians to measure end‑tidal CO₂ (EtCO₂) while delivering supplemental oxygen.
This dual function (oxygen delivery + CO₂ sampling) helps clinicians detect early signs of respiratory depression, hypoventilation, or airway issues. Available in adult, paediatric and ophthalmic sizes.
Ongoing Portfolio Expansion Across Specialties
Recent updates to the Mediplus product ecosystem demonstrate continued investment across Anaesthesia, Gynaecology, Urology, and Operating Room categories, reinforcing the company’s position as a leader in patient‑centric medical devices.
Global Marketing Director James Urie reinforces this direction:
“Mediplus maintains a strong development pipeline and continually seeks new ideas to stay at the forefront of clinical practice and medical technology.”
Manufacturing, Quality, Sustainability and Global Reach
Mediplus opened its own dedicated manufacturing facility in 2015, supporting its long‑term goal of full in‑house production, stringent quality control and driving increased sustainability.
The company holds:
• ISO 13485 certification
• CE Marking
• FDA approvals across a growing number of products
• ISO 14001 certification
• CSRA silver accreditation
Mediplus devices are now used throughout Europe, the USA, Canada, South America, Australia, New Zealand and the Far East.
Awards and Industry Recognition
Mediplus is a two‑time recipient of the Queen’s Award, receiving:
• Queen’s Award for Enterprise (International Trade) – 2013
• Queen’s Award for Enterprise (Innovation) – 2017
These achievements highlight the company’s commitment to excellence and global growth.