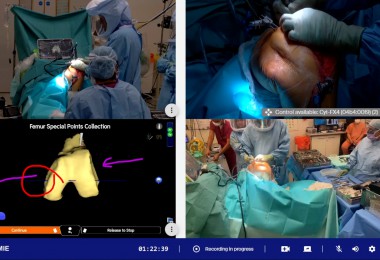
Mark Chapman, interim director of medical technology and digital evaluation, National Institute for Health and Care Excellence (NICE), highlights the tailored support available to the international medical technology industry, from product development to market access.
- For those that don’t know, what is NICE?
The role of NICE, simply, is to assess the value of a given health technology, as well as developing clinical guidelines that help inform care in the NHS. Fundamental to that, is striking a balance between the best care and value for money. It’s also important to note that we are independent: our decisions are rigorous, transparent and evidence based.
- What is your role within NICE?
With the emergence of a more hybrid way of working in the health and care environment, we acknowledge the important part that medtech, digital and diagnostics must play. My role had been newly created to bring strategic oversight to the great work that is already ongoing at NICE in these fields. I also aim to support some of the new, priority work that is being undertaken as part of our 5 year strategy, published in 2021.
- Do you work with companies that are not based in the UK, but are eager to bring their innovations to the UK?
Yes, we are keen to engage all industries wherever they are based if they have an appetite to enter the UK market. We aim to be a destination of choice for the leading innovations and innovators.
To help international companies who are looking to bring their products to the UK, NICE offers a range of early support services including: scientific advice, evidence gap analyses, health economic model review, and bespoke and confidential engagement meetings with relevant stakeholders to support market access.
These services are there to help small and large companies who are developing medicinal or medtech, digital and diagnostics products. We encourage any interested companies to get in touch with us to find out more.
- What is NICE International?
NICE International is our unique advisory service for health organisations, ministries and government agencies who want to promote evidence-based decision making in health and social care to improve outcomes in their countries.
NICE international initiates international partnerships to share best practice and world-leading NICE expertise. It also supports the development or advancement of local health technology assessment and guideline programmes. Our services support organisations to allocate limited healthcare resources, improve quality of care, reduce health inequalities and promote inclusivity and engagement.
- What can we expect to see from NICE in the future?
As I mentioned before, our 5-year strategy maps the long-term trends and challenges in health care and our overall direction of travel and ambitions. As the health and care landscape continues to change we need to be agile and continue to evolve the way we do things under the leadership of our new Chief Executive, Dr Sam Roberts. We know we can’t do everything. Therefore, we are focusing on those areas where NICE is internationally acknowledged as the expert in the system, and that provide the greatest benefit to people using health and care services.
We will do this through a focus on our priority areas. For example, we are working to provide earlier assessments of value to identify the most promising medtech, diagnostics and digital products - to support them to develop the further evidence required. Additionally, we aim to expand our capacity for technology appraisal guidance for medicines by 20% through developing a more proportionate approach. The usefulness and usability of our guidelines will be improved by developing digital living guideline recommendations in a non-COVID topic, with a new model of support for adoption of best practice. Finally, NICE is starting an internal transformation of the way we work, including our processes, technology and culture in order to support and deliver on the first 3 priorities.
You can find out more and subscribe to our newsletters, at www.nice.org.uk/life-sciences.